Challenge #3
| To develop a system where cell culture occurs in a contained chamber where contamination risks are minimized even when manipulated in non-classified laboratories. The system should be provided sterile, ready to use and, when in use, it should protect the cell culture from contamination from the surrounding environment. |
 |
One of the major concerns in cell culture is contamination. Detecting biological contamination in a cell batch could, literally, turn a laboratory upside down.
Unless the contamination source is clearly identified and can be eliminated, the responsible of the lab will want to ensure complete elimination of the contaminating agent before proceeding with further cultures. This will involve cleaning and sterilisation of laboratories and equipment and, most likely, replacing consumables that might have been exposed to the contamination source.
As for the different sources of contamination when considering the cell culture device, we can distinguish between intrinsic sources, this is, contamination originated in the device itself. And extrinsic sources or contamination originated outside the device.
Intrinsic sources of contamination
When re-utilizing a culture chamber, the risk of contaminating coming batches with the content of previous batches is very high. It is not only potential leftovers of the previous batch, but also the unavoidable contamination that will follow when the cell harvest has finished and the culture chamber is open and exposed to the surrounding environment.
This risk of contamination is the reason for carrying out extensive cleaning of reused equipment and setting up expensive cleaning validation protocols. This cleaning comes with important investments in clean-in-place (CIP) and sterilization-in-place (SIP) equipment and recurring expenses in operating costs for energy, chemicals, pure water, and labour. By using single-use / ready-to-use cell culture devices, considerations regarding this intrinsic source of contamination are eliminated.
Several methods there exist for sterilising cell culture devices. Choosing one method or the other will depend upon several factors, for example, if the device is thermolabile, autoclaving is out of the question, but maybe ethylene oxide is an option. For plastic elements radiation is an appropriate method, but it is not a good option for metallic parts. After analysing available sterilisation methods and taking into consideration all four challenges faced by The Bolt-on Bioreactor project, we conclude that sterilisation by radiation of a device compatible with this sterilization method is the best alternative for the BoB to become the standard for adherent cell culture. Therefore the culture device will be provided sterile, thus eliminating intrinsic contamination sources.
However, even if the cell culture device is provided sterile and ready to use, extrinsic sources of contamination remain, and the design of the product must help users minimize the risk of contaminating the culture from external contamination sources. Besides, cells should be kept within the device to avoid contamination of the laboratory or other ancillary elements used during the cell culture process.
Extrinsic sources of contamination
When analysing extrinsic contamination sources, we find that they arise from any of the following process related activities:
| Introducing or withdrawing fluids to or from the culture chamber, such as gases or culture medium. |
| Setting up probes and sensors for measuring various process parameters. |
| Rotatable or moving elements traversing the walls of the culture chamber, such as the shaft of a impeller. |
| Handling of the culture device by personnel or by dedicated machines, either of them spilling contaminating agents on the device, including air-borne contaminants. |
Let us now have a look to the contamination risks related to each of these activities.
Introducing or withdrawing fluids
The fluid with the highest turnover rate within the culture chamber is the gaseous phase. Unless you are using only air, certified gases are normally purchased from a vendor. Different qualities are available.
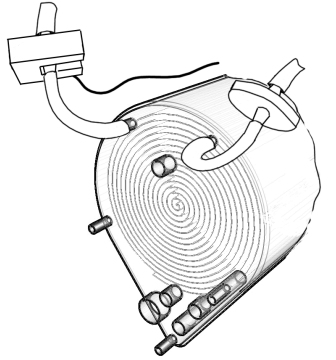
Some cell culturers rely on a pre-sterilised gas source as the only measure against contamination for their cultures, but this is highly uncommon. Most frequently a terminal sterilizing filter is placed just before the gas enters or leaves the culture chamber. This is a very efficient method to eliminate particulate contaminants born in the gas as long as the particle size of the contaminant is larger than the filter pore size. Usually, 0.45 um pore size filters are used for this purpose although larger and smaller pore size filters are available. Pore size and filter surface need be considered as they have an effect on pressure drop due to the resistance they oppose to gas flow. As for the exhaust filter, potential clogging of the filter should be taken into account, since venting gas has high relative humidity and there is a potential condensation risk on the filter membrane. Should filter clogging occur, pressure could build up within the culture chamber. A filter heating element solves this problem. The following figure is a squematic representation of gas inlet and oulet in the BoB showing terminal sterilising filters and a filter heater containing the filter in the gas outlet.
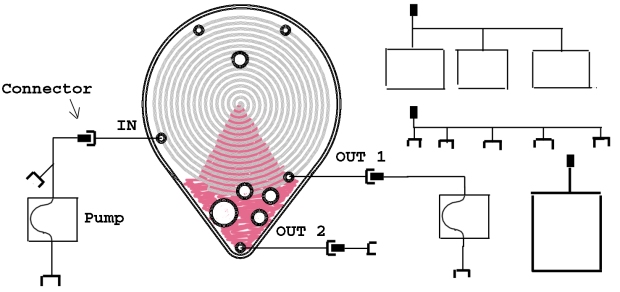
For liquid inlet and outlet during feeding and harvesting operations, sterilizing filters cannot be used due to the risks possed by the pressure necessary to operate the filter with liquids such as culture medium, and the undesired retention of components of interest on the filter membrane. Two major strategies remain to the cell culturer: 1) aseptically connecting every needed reservoir at the start of the culture process with enough volume to feed the cells throughout the whole process and collect all the waste and harvest or; 2) aseptically connecting and disconnecting reservoirs as needed during the process. Although the second strategy is more convenient to ensure that culture medium, waste, harvest and other liquid solutions employed during cell culture are stored at optimum conditions, it requires an appropriate system for aseptically connecting and disconnecting tubes. Fortunately, several alternatives are available for this purpose, and connectors standardization is spreading in the sector. Therefore we have chosen to increase the flexibility of the BoB by providing alternative connections to be chosen depending on the application of the culture and user preferences, and bearing in mind that set-up will be different if the product is in the supernatant - when producing antibodies, for example- or the product are the cells. The following figure shows possible designs to allow the user decide what what set-up is best for a given application. Different designs can be made available.
Seting up probes and sensors
Inserting probes on a culture vessel is always a risky operation. While sterile, ready to use tubing and filters are widely available, re-utilizable probes require careful sterilization prior to use. Besides, re-utilizable probes for measuring different parameters require different sterilization methods and have different set-up requirements. Seting up a probe can prove a delicate task when special connections are involved. Therefore, we have studied available options to reduce or eliminate contamination issues related to probes set-up.
First, we have analysed what control parameters are most widely used in adherent cell culture and then we have double checked by asking potential users on their opinion as for necessary control parameters. The results indicate that measurement of the following parameters should be incorporated by default in the BoB:
-rotation speed and direction
-medium replacement rate
-thermostating jacket temperature
-incoming gas temperature and flow
-gaseous phase CO2 concentration
-liquid phase dissolved oxygen
-liquid phase temperature
-liquid phase pH
Second we have studied available measuring methods for each of these parameters. And finally we have chosen the measuring method that minimizes contamination risk while providing sufficient precision and accuracy for each of these parameters. Happily, non-invasive measuring methods are available for all selected parameters. Dissolved oxygen, pH and CO2 concentration can be measured using optical probes or electrodes, the so-called optodes, where a small fluorescence-emitting reactive patch sends a luminous signal proportional to the analyte concentration. The luminous signal is transmitted trough the transparent wall of the culture vessel and received by an optic fiber that sends the signal to the transmitter for further processing. The following figure shows the proposed design for use of optodes and other probes in the BoB (1 shows a fluorescence-emitting sensitive patch to the left and the corresponding probe or optode to the right; 2 shows a temperature sensing probe being introduced in a pouch within the culture chamber and; 3 shows a user defined probe being introduced into the chamber)
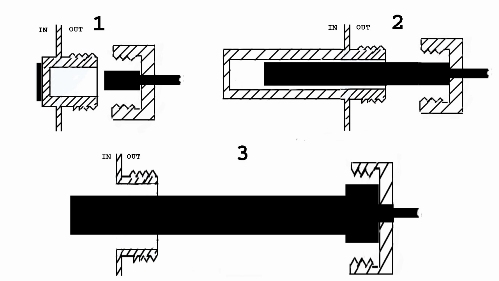
Temperature can be measured by introducing the probe inside a pouch that prolapses from the culture chamber wall towards inside the culture vessel, this way sensing the temperature inside the vessel in a non-invasive way. As for medium replacement rate and rotation speed and direction, both of these parameters are measured from the outside of the culture vessel.
Nevertheless, a spare port traversing the chamber wall and protected with a screw lid will be available for users with particular requirements.
Rotable or moving elements traversing the walls of the culture chamber
Having openings in the walls of the culture chamber is a constant concern regarding contamination. As we have seen, some of these openings are unavoidable. Such is the case for fluid inlets and outlets. Other openings that traditionally were necessary can be avoided, as is the case of non-invasive sensor elements. But if an opening is of concern, an opening where a rotating or moving element traversing the chamber wall is continuously in contact with both the inside and the outside of the culture chamber, is of major concern. This is the case of impeller shafts going through the wall of the culture chamber to provide agitation within the chamber. Work has been done to avoid the use of traversing shafts, and thus different agitation methods have been devised to be used in cell culture. We have grouped these methods in three broad groups, 1) agitation of the vessel, as used in rocking platforms agitating a bag partially filled with a cell suspension, or as used with rotating roller bottles; 2) bubling gas to agitate and aereate simultaneously and; 3) magnetic agitation, as used to rotate the impeller of some single-use bioreactors. A thorough review and experimentation with existing agitation methods where moving parts do not traverse the wall of the culture chamber, led us to the conclusion that none of them was adequate for The Bolt-on Bioreactor, since we want to avoid metallic elements within the culture chamber, we want a tight control of the rotatory movement, and we want a static culture chamber where tubes and cables can be connected and do not entangle due to the agitation of the chamber.
Following intensive design, we came up with an agitation system that perfectly fits the requirements of the BoB. The system is based on the mechanical coupling of plastic transfer balls within the culture chamber that acomodate within corresponding cavities between transfer balls on a driving disc outside the culture chamber, on the control unit. The balls or bearings within the culture chamber are separated from the bearings in the control unit by a flexible membrane, part of the chamber, that provides containment of the culture chamber without interfering with the mechanical coupling. Since the bearings inside the culture chamber are located in a disc integral with the rolled membrane where cells attach, the rotatory movement of the driving disc in the control unit conveys the rotatory movement to the rolled membrane, providing a system for the non-intrusive agitation of the rolled membrane within the culture vessel. The following figure is a schematic representation of the agitation system. A more detailed view of the movement transmission mechanism to the inside of the culture chamber is shown in the video at the end of this article.
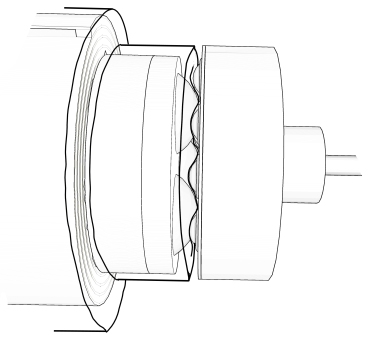
Handling of the culture device
Both, personnel and machinery are sources of contamination. While contamination carried by laboratory technicians is typically of biological origin, contamination originated on the equipment is most comonly related to aerosols and particles generated by heat and friction acting on lubricants and mechanical elements. On the design of The Bolt-on Bioreactor, we have managed to intercalate an isolating membrane between mechanical, moving elements and the environment containing the culture chamber. By doing so, it is now possible to locate control units in a room separated from the cell culture laboratory, facilitating maintainance tasks and reducing contamination risks from mechanical origin. The following figure shows a 9-chamber Multilab unit operated from within the lab while mechanic elements are segregated outside the room and separated by a flexible membrane (in blue).
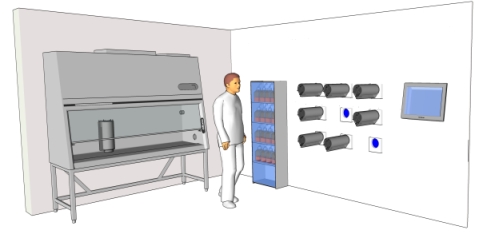
As for contamination originated on personnel, most critical operations are those where the inner space of the culture chamber or media containing reservoirs are exposed to laboratory environment. This will happen typically during bioreactor set-up at the start of the process and during operation whenever tubes need be connected or disconnected. Probes connection is also a risky operation. By reducing the number of times that tube manipulation is necessary, we reduce contamination opportunities. This is achieved through automation and continuous culture. But for those occassions where manual manipulation cannot be avoided, we rely on design features described earlier in this article.
Considering the possibility of contaminating the laboratory with the content of the culture device is also important. Situations that pose a risk of contaminating the laboratory are coincident with situations where the content of the culture device could become contaminated, therefore device design and accesories will take this fact into consideration. An exceptional situation may arise should the flexible membrane interacting with the control unit break for whatever reason. Strong materials will be used, but should this happen, the design of the device is such that culture medium feeding cells does not come in contact with the membrane and the cutlure chamber includes a reservoir big enough to acomodate all the liquid contained within the culture device, thus avoiding any chance of spilling chamber content into de lab.
The following video shows key design elements of the BoB that address Challenge #3.
© Copyright 2014, The BoB project
